Rather, the protein sequence of a humanized antibody is essentially identical to that of a human variant, despite the non-human origin of some of its complementarity-determining region (CDR) segments responsible for the ability of the antibody to bind to its target antigen.Ĭhimeric antibody names contain a -xi- stem. So, although the creation of an antibody chimera is normally undertaken to achieve a more human-like antibody (by replacing constant region of the mouse antibody with that from human) simple chimeras of this type are not usually referred to as humanized. 
Humanization is usually seen as distinct from the creation of a mouse-human antibody chimera. Human parts are shown in brown, non-human parts in blue. phage display) but essentially all are dependent on techniques that similarly allow the "insertion" or "swapping-out" of portions of the antibody molecule.ĭistinction from "chimeric antibody" Sketches of chimeric (top right), humanized (bottom left) and chimeric/humanized (bottom middle) monoclonal antibodies. Not all methods for deriving antibodies intended for human therapy require a humanization step (e.g. The alterations to antibody structure that are achieved in the humanization process are therefore all effectuated through techniques at the DNA level. 
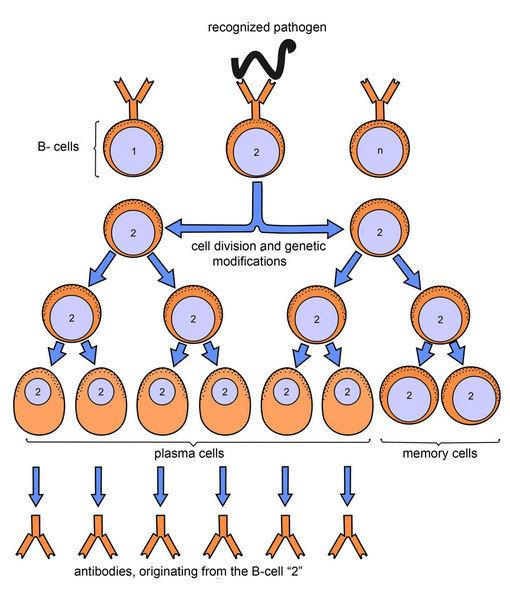
The step involving recombinant DNA provides an intervention point that can be readily exploited to alter the protein sequence of the expressed antibody. That is, gene segments capable of producing antibodies are isolated and cloned into cells that can be grown in a bioreactor such that antibody proteins produced from the DNA of the cloned genes can be harvested en masse. The humanization process takes advantage of the fact that production of monoclonal antibodies can be accomplished using recombinant DNA to create constructs capable of expression in mammalian cell culture. 
Use of recombinant DNA in humanization process This list covers many of the monoclonals developed for use in humans. There are other ways to develop monoclonal antibodies. The latter also have their protein sequences made more similar to human antibodies, but carry a larger stretch of non-human protein. Humanized antibodies are distinct from chimeric antibodies. The International Nonproprietary Names of humanized antibodies end in -zumab, as in omalizumab (see Nomenclature of monoclonal antibodies). The protein sequences of antibodies produced in this way are partially distinct from homologous antibodies occurring naturally in humans, and are therefore potentially immunogenic when administered to human patients (see also Human anti-mouse antibody). Humanization can be necessary when the process of developing a specific antibody involves generation in a non-human immune system (such as that in mice). The process of "humanization" is usually applied to monoclonal antibodies developed for administration to humans (for example, antibodies developed as anti-cancer drugs). Humanized antibodies are antibodies from non-human species whose protein sequences have been modified to increase their similarity to antibody variants produced naturally in humans. Antibodies from non-human species made nearly identical with human ones 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
